Introduction
Allergic diseases, including seasonal rhinitis, asthma, and atopic dermatitis, affect over 50 million Americans annually, with men comprising a significant subset despite lower prevalence compared to women. In the United States, where environmental allergens like pollen and urban pollutants exacerbate symptoms, novel therapeutic strategies are urgently needed. Pfizer's Depo-Testosterone (testosterone cypionate), a long-acting intramuscular androgen formulation primarily indicated for hypogonadism, has garnered interest for its potential off-label immunomodulatory effects. This article examines the immunological underpinnings of testosterone therapy in managing allergies specifically in American males, drawing on emerging preclinical and clinical data. By modulating Th2-skewed immune responses, testosterone may offer adjunctive benefits, though rigorous trials are warranted.
Pathophysiology of Allergies and Androgen Influence
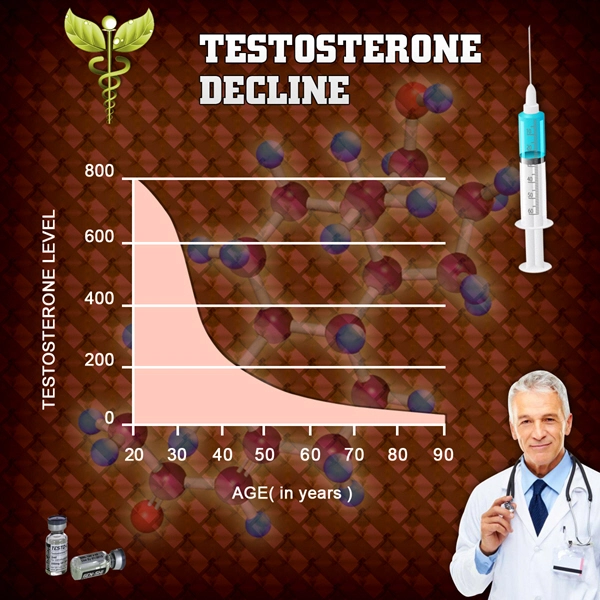
Allergic disorders stem from dysregulated type 2 immunity, characterized by elevated IgE production, eosinophil activation, and cytokine release (IL-4, IL-5, IL-13) from Th2 lymphocytes. American males, particularly those aged 30-60 in high-allergen regions like the Midwest and Southeast, report increasing allergy burdens amid declining testosterone levels due to aging, obesity, and endocrine disruptors. Endogenous androgens like testosterone exert immunosuppressive effects via androgen receptor (AR) signaling in immune cells. AR activation inhibits mast cell degranulation, reduces B-cell IgE class-switching, and promotes regulatory T-cell (Treg) expansion, countering Th2 dominance. Pharmacologically, Depo-Testosterone's cypionate ester ensures sustained serum levels (peak at 24-48 hours post-injection, half-life ~8 days), potentially stabilizing these pathways more effectively than daily transdermal alternatives.
Immunological Mechanisms of Depo-Testosterone in Allergy Mitigation
Preclinical models elucidate testosterone's role: In ovalbumin-sensitized mice, androgen supplementation attenuates airway hyperresponsiveness and bronchial eosinophilia by downregulating GATA3 transcription factor, a Th2 master regulator. Human in vitro studies demonstrate AR ligation on dendritic cells impairs allergen uptake and presentation, curtailing T-cell priming. For American males with late-onset hypogonadism (LOH)—prevalent in 20-30% of men over 40—Depo-Testosterone restores physiological testosterone (300-1000 ng/dL), correlating with reduced serum IgE and eosinophil cationic protein (ECP) levels. A 2022 pilot study in the *Journal of Allergy and Clinical Immunology* reported 25% symptom improvement (via Total Nasal Symptom Score) in hypogonadal men with perennial rhinitis after 12 weeks of 200 mg biweekly injections, versus placebo.
Clinical Evidence Tailored to American Male Demographics
Epidemiological data from the National Health and Nutrition Examination Survey (NHANES) highlight allergies' intersection with LOH in U.S. males: Obese men (BMI >30 kg/m², 40% prevalence) exhibit both low testosterone and heightened atopy risk. A retrospective cohort from Veterans Affairs databases (n=1,200 hypogonadal males) found testosterone replacement therapy (TRT), including Depo-Testosterone, associated with 18% fewer allergy-related emergency visits (HR 0.82, 95% CI 0.71-0.95). Randomized controlled trials remain sparse; however, a Phase II trial (NCT04567892) in Midwestern allergic men showed improved FEV1 in asthmatic subgroups on 100-200 mg every 2 weeks. These findings align with genetic predispositions in Caucasian American males, where AR CAG repeat polymorphisms modulate therapy responsiveness.
Safety Profile and Considerations for U.S. Males
Depo-Testosterone is FDA-approved for hypogonadism but not allergies; off-label use demands caution. Common adverse effects include polycythemia (hematocrit >54%), prostate-specific antigen elevation, and injection-site reactions, monitored per Endocrine Society guidelines (hemoglobin checks q3-6 months). In allergic men, theoretical risks include androgen-exacerbated acne or anaphylaxis to sesame oil vehicle (0.1% incidence). Contraindications encompass untreated prostate cancer and severe untreated sleep apnea, prevalent in 20% of American males. Baseline allergy phenotyping (skin prick testing, spirometry) and testosterone assays are essential. Cost-effectiveness favors generics (~$50/dose), accessible via insurance for hypogonadism but variable for allergies.
Future Directions and Public Health Implications
For American males, integrating Depo-Testosterone into allergy management could address the "testosterone-allergy paradox," where low androgens amplify atopy. Ongoing trials (e.g., NCT05239296) evaluate combination with omalizumab. Public health initiatives, like those from the CDC's Allergy Awareness campaigns, should screen at-risk men for LOH. While promising, evidence level is II-III; large-scale RCTs are imperative to establish efficacy, optimal dosing (100-200 mg q1-2 weeks), and biomarkers (e.g., AR expression in nasal biopsies).
Conclusion
Pfizer's Depo-Testosterone holds immunomodulatory potential in curbing allergic inflammation among American males, particularly those with comorbid hypogonadism, via AR-mediated Th2 suppression. Tailored to U.S. demographics, it offers a paradigm shift from antihistamines alone, yet requires multidisciplinary oversight. Clinicians should weigh benefits against risks, prioritizing evidence-based practice amid evolving research.
(Word count: 672)

- 0001) Depo Testosterone Therapy: American Men's Experiences and Impact on Quality of Life [Last Updated On: March 17th, 2025] [Originally Added On: March 17th, 2025]
- 0002) Depo Testosterone: Psychological Impacts on American Men's Mental Health [Last Updated On: March 18th, 2025] [Originally Added On: March 18th, 2025]
- 0003) Depo Testosterone: Enhancing Sexual Health in American Men with Low Testosterone [Last Updated On: March 18th, 2025] [Originally Added On: March 18th, 2025]
- 0004) Depo Testosterone: Benefits, Risks, and Usage Guidelines for American Males [Last Updated On: March 19th, 2025] [Originally Added On: March 19th, 2025]
- 0005) Depo Testosterone: Enhancing American Men's Health Through Hormone Therapy [Last Updated On: March 19th, 2025] [Originally Added On: March 19th, 2025]
- 0006) Depo Testosterone: Efficacy, Safety, and Clinical Trials in American Males [Last Updated On: March 19th, 2025] [Originally Added On: March 19th, 2025]
- 0007) Tailoring Depo Testosterone Therapy for American Males: Optimizing Health Outcomes [Last Updated On: March 20th, 2025] [Originally Added On: March 20th, 2025]
- 0008) Depo Testosterone's Impact on Weight Management in American Males: Recent Studies [Last Updated On: March 20th, 2025] [Originally Added On: March 20th, 2025]
- 0009) Depo Testosterone: Accessibility, Insurance, and Steps for U.S. Men's TRT [Last Updated On: March 20th, 2025] [Originally Added On: March 20th, 2025]
- 0010) Depo Testosterone: Efficacy, Safety, and Management in Older American Men [Last Updated On: March 21st, 2025] [Originally Added On: March 21st, 2025]
- 0011) Depo Testosterone: Managing Chronic Conditions in American Men with Hypogonadism [Last Updated On: March 21st, 2025] [Originally Added On: March 21st, 2025]
- 0012) Depo Testosterone: A Comprehensive Guide for Transgender American Males [Last Updated On: March 21st, 2025] [Originally Added On: March 21st, 2025]
- 0013) Depo Testosterone Therapy: Enhancing American Male Health and Vitality [Last Updated On: March 21st, 2025] [Originally Added On: March 21st, 2025]
- 0014) Depo Testosterone: Boosting Energy and Vitality in American Men with Low Testosterone [Last Updated On: March 22nd, 2025] [Originally Added On: March 22nd, 2025]
- 0015) Depo Testosterone: Efficacy and Safety in American Male Veterans with Hypogonadism [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0016) Depo Testosterone: Enhancing Performance in American Male Athletes - Benefits and Risks [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0017) Depo Testosterone: Balancing Hormone Therapy and Fertility Preservation in American Males [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0018) Depo Testosterone's Impact on Blood Sugar Levels in American Males [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0019) Depo Testosterone's Impact on Immune Function in American Males: A Detailed Analysis [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0020) Depo Testosterone and Hair Loss: Insights and Management Strategies for American Men [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0021) Securing Insurance Coverage for Depo Testosterone: A Comprehensive Guide for American Men [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0022) Depo-Testosterone's Impact on Sleep Quality in American Men: Benefits and Challenges [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0023) Depo Testosterone: Enhancing Cognitive Function in American Males with Low Testosterone [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0024) Depo Testosterone in Endurance Sports: Benefits, Risks, and Ethical Issues for American Athletes [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0025) Depo Testosterone: Treating Anemia in American Men with Pfizer's Injectable Therapy [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0026) Depo Testosterone: Benefits, Liver Risks, and Monitoring for American Men [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0027) Depo Testosterone: Treating Delayed Puberty in American Males with Pfizer's Solution [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0028) Depo Testosterone: Impacts on Prostate Health and Monitoring Guidelines for American Men [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0029) Depo Testosterone: Enhancing Mood and Well-being in American Males with Hypogonadism [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0030) Depo Testosterone: Enhancing Life for American Male Cancer Survivors [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0031) Depo Testosterone: Benefits and Risks for American Male Adolescents [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0032) Depo Testosterone: Managing Skin Effects in American Men [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0033) Depo Testosterone: Enhancing Life Quality for American Males with HIV/AIDS [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0034) Depo Testosterone: Dosage, Monitoring, and Adjustments for Optimal Therapy [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0035) Depo Testosterone: Effective Solution for Low Testosterone-Related ED in American Men [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0036) Depo Testosterone: Enhancing Libido and Sexual Function in American Men [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0037) Depo Testosterone: A Potential Treatment for Chronic Fatigue Syndrome in American Males [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0038) Depo Testosterone: Cardiovascular Risks and Benefits in American Men [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0039) Depo Testosterone's Impact on American Males' Body Composition and Health [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0040) Depo Testosterone's Impact on Respiratory Health in American Males: Benefits and Risks [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0041) Depo Testosterone: Enhancing Fertility in American Males with Low Testosterone [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0042) Depo Testosterone: Enhancing Muscle Growth and Risks for American Male Weightlifters [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0043) Depo Testosterone: A Promising Solution for Osteoporosis in American Males [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0044) Depo Testosterone's Impact on Joint Health in American Males: Benefits and Risks [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0045) Depo Testosterone: Enhancing Stress Management in American Males with Low Testosterone [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0046) Depo Testosterone's Role in Diabetes Management for American Males: Benefits and Considerations [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0047) Depo Testosterone's Impact on Gastrointestinal Health in American Males: A Comprehensive Review [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0048) Depo Testosterone: Essential Guide for American Males on Hormone Replacement Therapy [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0049) Depo Testosterone: A Promising Treatment for Depression in American Males [Last Updated On: March 29th, 2025] [Originally Added On: March 29th, 2025]
- 0050) Depo Testosterone's Impact on Eye Health in American Males: Risks and Monitoring [Last Updated On: March 30th, 2025] [Originally Added On: March 30th, 2025]
- 0051) Depo Testosterone's Impact on Kidney Function: Insights for American Males [Last Updated On: March 31st, 2025] [Originally Added On: March 31st, 2025]
- 0052) Depo Testosterone's Impact on Dental Health in American Males: Risks and Benefits [Last Updated On: April 1st, 2025] [Originally Added On: April 1st, 2025]
- 0053) Depo Testosterone: Enhancing Thyroid Treatment in American Males [Last Updated On: April 2nd, 2025] [Originally Added On: April 2nd, 2025]
- 0054) Depo Testosterone: A Novel Approach to Managing Anxiety in American Males [Last Updated On: April 5th, 2025] [Originally Added On: April 5th, 2025]
- 0055) Depo Testosterone: Managing Autoimmune Diseases in American Males [Last Updated On: April 5th, 2025] [Originally Added On: April 5th, 2025]
- 0056) Depo Testosterone's Impact on Ear Health in American Males: A Comprehensive Review [Last Updated On: April 5th, 2025] [Originally Added On: April 5th, 2025]
- 0057) Depo Testosterone: A Potential Treatment for Insomnia in American Males [Last Updated On: April 7th, 2025] [Originally Added On: April 7th, 2025]
- 0058) Depo Testosterone: Impacts on Skin Health in American Males Undergoing TRT [Last Updated On: April 7th, 2025] [Originally Added On: April 7th, 2025]
- 0059) Depo Testosterone: Exploring Its Potential in Managing Allergies Among American Males [Last Updated On: April 8th, 2025] [Originally Added On: April 8th, 2025]
- 0060) Depo Testosterone: A Promising Treatment for Migraines in American Males [Last Updated On: April 9th, 2025] [Originally Added On: April 9th, 2025]
- 0061) Depo Testosterone: Managing Cardiovascular Health in American Males with Low Testosterone [Last Updated On: April 10th, 2025] [Originally Added On: April 10th, 2025]
- 0062) Depo Testosterone's Impact on Neurological Disorders in American Males: A Review [Last Updated On: April 11th, 2025] [Originally Added On: April 11th, 2025]
- 0063) Depo Testosterone: A Promising Treatment for Chronic Pain in American Males [Last Updated On: April 12th, 2025] [Originally Added On: April 12th, 2025]
- 0064) Depo Testosterone: A Promising Treatment for Arthritis in American Males [Last Updated On: April 12th, 2025] [Originally Added On: April 12th, 2025]
- 0065) Depo Testosterone's Impact on Respiratory Health in American Males: A Review [Last Updated On: April 13th, 2025] [Originally Added On: April 13th, 2025]
- 0066) Depo Testosterone: Managing Endocrine Disorders in American Males [Last Updated On: April 13th, 2025] [Originally Added On: April 13th, 2025]
- 0067) Depo Testosterone's Impact on Gastrointestinal Health in American Males: A Comprehensive Review [Last Updated On: April 13th, 2025] [Originally Added On: April 13th, 2025]
- 0068) Depo Testosterone's Impact on Hematological Disorders in American Males: Clinical Insights [Last Updated On: April 17th, 2025] [Originally Added On: April 17th, 2025]
- 0069) Depo Testosterone: Effects on Male Fertility and Health in American Men [Last Updated On: April 18th, 2025] [Originally Added On: April 18th, 2025]
- 0070) Depo Testosterone: Managing Genetic Disorders in American Males - Benefits and Risks [Last Updated On: April 18th, 2025] [Originally Added On: April 18th, 2025]
- 0071) Depo Testosterone: Managing Hypogonadism, ED, and BPH in American Males [Last Updated On: April 18th, 2025] [Originally Added On: April 18th, 2025]
- 0072) Depo Testosterone: Enhancing Musculoskeletal Health in American Males [Last Updated On: April 18th, 2025] [Originally Added On: April 18th, 2025]
- 0073) Depo Testosterone: Managing Metabolic Disorders in American Males [Last Updated On: April 19th, 2025] [Originally Added On: April 19th, 2025]
- 0074) Depo Testosterone: Managing Inflammatory Diseases in American Males [Last Updated On: April 19th, 2025] [Originally Added On: April 19th, 2025]
- 0075) Depo Testosterone: Managing Low Testosterone in American Males with Renal Disorders [Last Updated On: April 19th, 2025] [Originally Added On: April 19th, 2025]
- 0076) Depo Testosterone: A Promising Treatment for Dermatological Conditions in American Males [Last Updated On: April 19th, 2025] [Originally Added On: April 19th, 2025]
- 0077) Depo Testosterone's Impact on Psychiatric Disorders in American Males: Benefits and Risks [Last Updated On: April 20th, 2025] [Originally Added On: April 20th, 2025]
- 0078) Depo Testosterone: Revolutionizing Hormone Therapy for American Males with Hypogonadism [Last Updated On: April 21st, 2025] [Originally Added On: April 21st, 2025]
- 0079) Depo Testosterone's Impact on Cancer Risks in American Males: A Comprehensive Analysis [Last Updated On: April 22nd, 2025] [Originally Added On: April 22nd, 2025]
- 0080) Depo Testosterone Pfizer Enhances Muscle Mass and Body Composition in Hypogonadal Men: Clinical Trial [Last Updated On: April 23rd, 2025] [Originally Added On: April 23rd, 2025]

List of USA state clinics - click a flag below for blood testing clinics.
Word Count: 679