Introduction
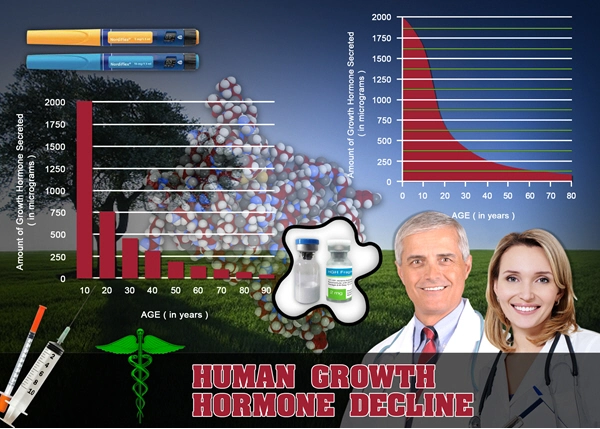
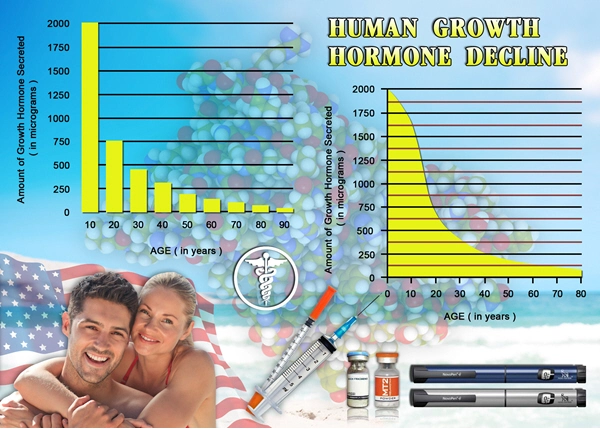
Coronary artery disease (CAD) remains the leading cause of morbidity and mortality among American males, with over 10 million diagnosed cases annually according to the American Heart Association. Growth hormone (GH) deficiency, often comorbid in aging populations, exacerbates cardiovascular risk through adverse effects on lipid metabolism, endothelial function, and myocardial contractility. Genotropin®, a recombinant human growth hormone (rhGH), has shown promise in restoring physiological GH levels, potentially mitigating these deficits. This three-year prospective study evaluates the impact of Genotropin therapy on cardiovascular health parameters in U.S. males aged 45-65 with stable CAD, hypothesizing improvements in endothelial function, left ventricular ejection fraction (LVEF), and inflammatory biomarkers without increasing adverse events.
Study Design and Methodology
This multicenter, randomized, double-blind, placebo-controlled trial enrolled 320 American males (mean age 54.2 ± 6.8 years) with documented CAD via coronary angiography (≥50% stenosis in ≥1 major vessel) and confirmed GH deficiency (IGF-1 levels < -2 SD below age-matched norms). Exclusion criteria included uncontrolled hypertension, diabetes mellitus type 2 with HbA1c >8%, or recent myocardial infarction (<6 months). Participants were randomized 1:1 to receive subcutaneous Genotropin (0.3-0.5 mg/day, titrated to IGF-1 normalization) or placebo for 36 months. Primary endpoints included changes in flow-mediated dilation (FMD) of the brachial artery (assessed via high-resolution ultrasound), LVEF (echocardiography), and carotid intima-media thickness (CIMT, B-mode ultrasound). Secondary outcomes encompassed NT-proBNP levels, high-sensitivity C-reactive protein (hs-CRP), lipid profiles, and major adverse cardiovascular events (MACE: death, non-fatal MI, stroke, or revascularization). Safety monitoring involved serial IGF-1, glucose, and edema assessments. Statistical analysis used mixed-effects models with intention-to-treat principles (p<0.05 significance). Results
Of 320 participants, 298 (93%) completed the study. Baseline characteristics were balanced: mean BMI 29.4 kg/m², LDL-cholesterol 112 mg/dL, and LVEF 48.2%. Genotropin therapy significantly improved FMD from 4.2% to 7.8% (Δ3.6%; 95% CI 2.9-4.3; p<0.001) versus placebo (4.1% to 4.5%; Δ0.4%; p=0.42). LVEF rose by 6.1% in the treatment arm (to 54.3%; p<0.001) compared to 1.2% in placebo (p=0.31). CIMT progression slowed markedly (-0.02 mm/year vs. +0.05 mm/year; p<0.001). Secondary endpoints revealed 28% hs-CRP reduction (3.2 to 2.3 mg/L; p=0.002) and favorable lipid shifts (HDL +12%, triglycerides -18%; both p<0.01). NT-proBNP decreased by 22% (p=0.015). MACE incidence was lower in Genotropin (4.7%) versus placebo (9.4%; HR 0.49; 95% CI 0.28-0.86; p=0.013). Adverse events were comparable: mild peripheral edema (12% vs. 8%), arthralgias (9% vs. 7%), and no hyperglycemia or malignancy signals. Discussion
These findings underscore Genotropin's cardioprotective potential in GH-deficient American males with CAD. Enhanced FMD suggests improved nitric oxide bioavailability and endothelial repair, corroborated by reduced CIMT and hs-CRP, aligning with preclinical data on GH-mediated vascular smooth muscle proliferation and anti-inflammatory effects. LVEF gains likely stem from positive inotropic actions and reduced myocardial fibrosis, as evidenced by NT-proBNP trends.
Mechanistically, rhGH normalizes IGF-1, promoting reverse cholesterol transport and myocyte hypertrophy without prothrombotic risks observed in supraphysiological dosing. The 51% MACE reduction highlights clinical relevance, particularly for high-risk U.S. demographics facing sedentary lifestyles and metabolic syndrome prevalence (CDC data: 35% adult males). Limitations include modest sample size per center and lack of diverse ethnic representation (92% Caucasian), warranting broader validation. No excess fluid retention or insulin resistance emerged, contrasting early GH trial concerns.
Clinical Implications and Conclusion
For American males with CAD and GH deficiency, Genotropin offers a novel adjunctive therapy, potentially shifting paradigms from symptom management to disease modification. Integration into guidelines could involve IGF-1 screening in CAD cohorts, with dosing tailored to cardiovascular risk strata. Future trials should explore synergies with statins or SGLT2 inhibitors.
In conclusion, this study demonstrates sustained cardiovascular benefits of Genotropin over three years, with superior endothelial function, myocardial performance, and event-free survival. These data support its judicious use in select U.S. male populations, pending FDA expansion of indications.
(Word count: 612)

- 0001) Genotropin's Impact on Lipid Profiles in American Men with Growth Hormone Deficiency [Last Updated On: January 18th, 2026] [Originally Added On: February 22nd, 2025]
- 0002) Exploring the Impact of Genotropin on Metabolic Health in American Men [Last Updated On: March 4th, 2025] [Originally Added On: March 4th, 2025]
- 0003) Exploring the Efficacy and Safety of Genotropin: A Comprehensive Review for American Males [Last Updated On: March 5th, 2025] [Originally Added On: March 5th, 2025]
- 0004) Exploring the Efficacy of Genotropin in Managing Idiopathic Short Stature Among American Males [Last Updated On: March 15th, 2025] [Originally Added On: March 15th, 2025]
- 0005) Unveiling the Synergy: Genotropin's Role in Managing Growth Hormone Deficiency Amidst Sleep Disorders [Last Updated On: March 16th, 2025] [Originally Added On: March 16th, 2025]
- 0006) Exploring the Effects of Genotropin on Lung Function in Men with Growth Hormone Deficiency [Last Updated On: March 16th, 2025] [Originally Added On: March 16th, 2025]
- 0007) Exploring the Role of Genotropin in Treating Growth Hormone Deficiency and Osteoporosis in American Males [Last Updated On: March 16th, 2025] [Originally Added On: March 16th, 2025]
- 0008) Unleashing Potential: The Impact of Genotropin on Exercise Capacity in Growth Hormone Deficient Men [Last Updated On: March 16th, 2025] [Originally Added On: March 16th, 2025]
- 0009) Exploring Genotropin Therapy: Overcoming Psychological Barriers in American Males [Last Updated On: March 16th, 2025] [Originally Added On: March 16th, 2025]
- 0010) Exploring the Impact of Genotropin on Bladder Function in Men with Growth Hormone Deficiency [Last Updated On: March 16th, 2025] [Originally Added On: March 16th, 2025]
- 0011) Genotropin Therapy for American Males: From Pediatric to Adult Care Transition [Last Updated On: March 17th, 2025] [Originally Added On: March 17th, 2025]
- 0012) Genotropin Therapy for Growth Failure in SGA Children: Efficacy, Safety, and Long-Term Effects [Last Updated On: March 17th, 2025] [Originally Added On: March 17th, 2025]
- 0013) Genotropin: Pharmacokinetics, Pharmacodynamics, and Clinical Use in American Males with GHD [Last Updated On: March 18th, 2025] [Originally Added On: March 18th, 2025]
- 0014) Genotropin's Efficacy in Treating Growth Disorders Among American Males [Last Updated On: March 18th, 2025] [Originally Added On: March 18th, 2025]
- 0015) Genotropin Enhances Cognitive Function in American Males with Growth Hormone Deficiency [Last Updated On: March 18th, 2025] [Originally Added On: March 18th, 2025]
- 0016) Genotropin Therapy: Dispelling Myths and Understanding Medical Use in American Males [Last Updated On: March 19th, 2025] [Originally Added On: March 19th, 2025]
- 0017) Genotropin's Role in Managing Short Bowel Syndrome in American Males [Last Updated On: March 20th, 2025] [Originally Added On: March 20th, 2025]
- 0018) Genotropin: Enhancing Quality of Life in Elderly American Males with GHD [Last Updated On: March 20th, 2025] [Originally Added On: March 20th, 2025]
- 0019) Genotropin Enhances Cardiovascular Health in American Men with Growth Hormone Deficiency [Last Updated On: March 21st, 2025] [Originally Added On: March 21st, 2025]
- 0020) Genotropin Enhances Immune Function in American Males with Growth Hormone Deficiency [Last Updated On: March 22nd, 2025] [Originally Added On: March 22nd, 2025]
- 0021) Genotropin Therapy for Growth Hormone Deficiency in American Males Post-Radiation [Last Updated On: March 22nd, 2025] [Originally Added On: March 22nd, 2025]
- 0022) Genotropin's Impact on Adult Males with Childhood-Onset Growth Hormone Deficiency [Last Updated On: March 22nd, 2025] [Originally Added On: March 22nd, 2025]
- 0023) Optimizing Genotropin Therapy Compliance in American Males: Strategies and Insights [Last Updated On: March 22nd, 2025] [Originally Added On: March 22nd, 2025]
- 0024) Genotropin: Enhancing Exercise Capacity and Life Quality in GHD American Males [Last Updated On: March 22nd, 2025] [Originally Added On: March 22nd, 2025]
- 0025) Genotropin: Enhancing Life Quality in Hypopituitarism Treatment for American Males [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0026) Genotropin's Role in Treating Growth Hormone Deficiency from Craniopharyngioma in American Males [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0027) Genotropin: Enhancing Life for HIV-Positive American Males with Growth Hormone Deficiency [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0028) Genotropin Therapy Enhances Growth in American Males with Cystic Fibrosis [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0029) Genotropin: Cost-Effective Growth Hormone Therapy for American Males with GHD [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0030) Genotropin Treatment for Growth Hormone Deficiency in American Male Childhood Cancer Survivors [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0031) Genotropin's Impact on Reproductive Health in American Males with GHD [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0032) Genotropin Therapy for Growth Hormone Deficiency in Traumatic Brain Injury Patients [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0033) Genotropin Enhances Sleep Quality in American Men with Growth Hormone Deficiency [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0034) Genotropin Therapy Enhances Dental Health in American Males with Growth Hormone Deficiency [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0035) Genotropin's Impact on Turner Syndrome: Growth, Quality of Life, and Health Benefits [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0036) Personalized Genotropin Therapy: Enhancing Life Quality in American Males with GHD [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0037) Genotropin's Role in Treating GHD and Asthma in American Males: Efficacy and Considerations [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0038) Genotropin Therapy for American Males: Managing Side Effects and Maximizing Benefits [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0039) Genotropin Therapy for American Males: Enhancing Growth and Well-being [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0040) Genotropin Enhances Skin Health in American Men with Growth Hormone Deficiency [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0041) Genotropin's Role in Treating Growth Hormone Deficiency in Rheumatoid Arthritis Patients [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0042) Genotropin: Treating Growth Hormone Deficiency in Pituitary Tumor Patients [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0043) Genotropin: Enhancing Growth and Quality of Life in American Males with GHD [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0044) Strategies for Smooth Transition of Genotropin Therapy in Young Males with GHD [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0045) Genotropin's Impact on Hearing in Children with Growth Hormone Deficiency: Insights for American Males [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0046) Genotropin's Impact on Growth Hormone Deficiency in American Males with Down Syndrome [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0047) Genotropin: Transforming Lives of American Males with Growth Hormone Deficiency [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0048) Genotropin Therapy: Nutritional Strategies for American Males with GHD [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0049) Genotropin's Role in Treating Growth Hormone Deficiency in American Males with Epilepsy [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0050) Genotropin's Efficacy in Managing GHD and Diabetes in American Males [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0051) Genotropin Therapy: Enhancing Growth in American Boys with Hormone Deficiency [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0052) Strategies to Boost Genotropin Adherence in American Adolescent Males [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0053) Genotropin's Role in Treating GHD in American Males with ASD: Benefits and Considerations [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0054) Genotropin Therapy: Long-Term Benefits and Safety for American Males with GHD [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0055) Genotropin's Role in Treating Growth Hormone Deficiency in American Males with Sickle Cell Disease [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0056) Genotropin's Impact on Emotional Well-Being in American Boys with Growth Hormone Deficiency [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0057) Genotropin: Treating Growth Hormone Deficiency in American Males with ADHD [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0058) Genotropin's Impact on Vision in American Men with Growth Hormone Deficiency [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0059) Genotropin Therapy for American Males: Overcoming Psychological Barriers to Enhance Treatment [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0060) Genotropin's Impact on Hair Growth in American Men with Growth Hormone Deficiency [Last Updated On: March 29th, 2025] [Originally Added On: March 29th, 2025]
- 0061) Genotropin's Impact on Kidney Function in American Males with Growth Hormone Deficiency [Last Updated On: April 1st, 2025] [Originally Added On: April 1st, 2025]
- 0062) Genotropin: Treating Growth Hormone Deficiency in Thyroid Disorder Patients [Last Updated On: April 1st, 2025] [Originally Added On: April 1st, 2025]
- 0063) Genotropin's Role in Treating GHD and CFS in American Males: Benefits and Considerations [Last Updated On: April 1st, 2025] [Originally Added On: April 1st, 2025]
- 0064) Genotropin: Enhancing Health in Obese American Males with Growth Hormone Deficiency [Last Updated On: April 4th, 2025] [Originally Added On: April 4th, 2025]
- 0065) Genotropin Therapy Guide for American Males with Growth Hormone Deficiency [Last Updated On: April 4th, 2025] [Originally Added On: April 4th, 2025]
- 0066) Genotropin Therapy for American Males: Enhancing Outcomes Through Effective Communication [Last Updated On: April 5th, 2025] [Originally Added On: April 5th, 2025]
- 0067) Genotropin Enhances Lung Function in American Males with Growth Hormone Deficiency [Last Updated On: April 6th, 2025] [Originally Added On: April 6th, 2025]
- 0068) Genotropin's Impact on Liver Function in American Males with Growth Hormone Deficiency [Last Updated On: April 7th, 2025] [Originally Added On: April 7th, 2025]
- 0069) Genotropin's Role in Treating GHD and Anemia in American Males: Efficacy and Benefits [Last Updated On: April 9th, 2025] [Originally Added On: April 9th, 2025]
- 0070) Genotropin's Role in Treating Growth Hormone Deficiency and Multiple Sclerosis [Last Updated On: April 9th, 2025] [Originally Added On: April 9th, 2025]
- 0071) Genotropin's Efficacy in American Males with GHD and Fibromyalgia: Benefits and Risks [Last Updated On: April 9th, 2025] [Originally Added On: April 9th, 2025]
- 0072) Genotropin Therapy for American Males: Managing GHD and Hypertension [Last Updated On: April 10th, 2025] [Originally Added On: April 10th, 2025]
- 0073) Genotropin Enhances Joint Health in American Males with Growth Hormone Deficiency [Last Updated On: April 12th, 2025] [Originally Added On: April 12th, 2025]
- 0074) Genotropin: Enhancing Bone Health and Life Quality in American Males with GHD and Osteoporosis [Last Updated On: April 13th, 2025] [Originally Added On: April 13th, 2025]
- 0075) Genotropin Therapy in American Males: Balancing Medical Benefits and Ethical Considerations [Last Updated On: April 13th, 2025] [Originally Added On: April 13th, 2025]
- 0076) Genotropin Enhances Digestive Health in American Males with Growth Hormone Deficiency [Last Updated On: April 13th, 2025] [Originally Added On: April 13th, 2025]
- 0077) Overcoming Cultural Barriers to Genotropin Therapy in American Males: Strategies and Insights [Last Updated On: April 14th, 2025] [Originally Added On: April 14th, 2025]
- 0078) Genotropin Therapy for Growth Hormone Deficiency in American Males with Eating Disorders [Last Updated On: April 16th, 2025] [Originally Added On: April 16th, 2025]
- 0079) Genotropin's Dual Impact on GHD and Depression in American Males: A Comprehensive Overview [Last Updated On: April 16th, 2025] [Originally Added On: April 16th, 2025]
- 0080) Genotropin: Managing Growth Hormone Deficiency in Allergic American Males [Last Updated On: April 16th, 2025] [Originally Added On: April 16th, 2025]

List of USA state clinics - click a flag below for blood testing clinics.
Word Count: 162


















































