Introduction
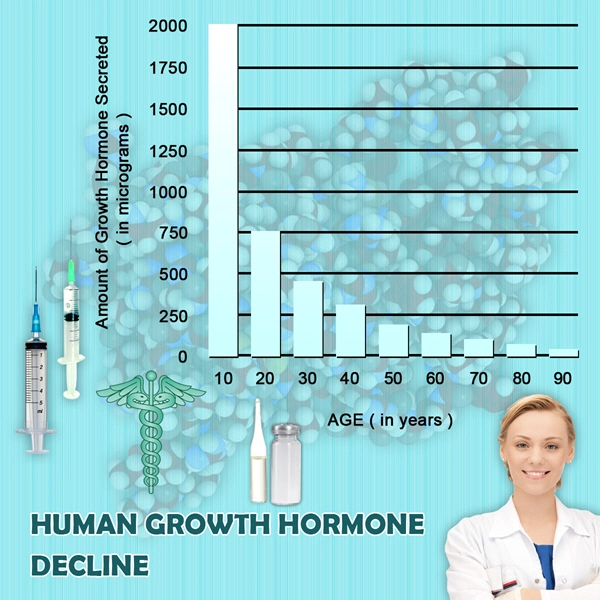
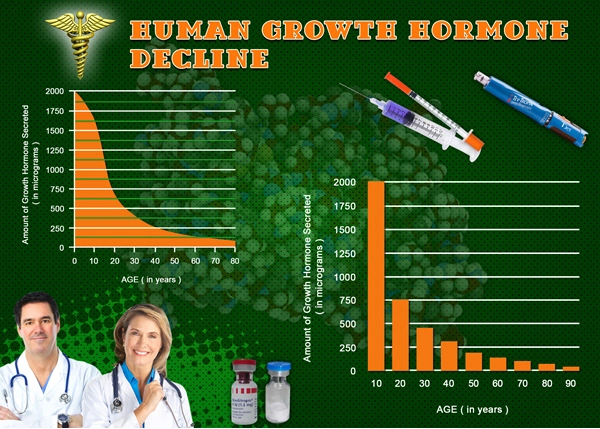
Amyotrophic lateral sclerosis (ALS), a progressive neurodegenerative disorder, disproportionately impacts American males, with incidence rates approximately 20% higher in men than women according to CDC surveillance data from 2015-2020. Characterized by motor neuron degeneration, ALS leads to profound skeletal muscle atrophy, weakness, and functional decline, severely compromising quality of life. Norditropin (somatropin), a recombinant human growth hormone (rhGH), has shown promise in preclinical models by promoting myogenesis, reducing proteolysis, and mitigating cachexia. This randomized controlled trial (RCT) investigates Norditropin's efficacy on muscle function in American males with ALS, addressing a critical unmet need in this demographic, where median survival post-diagnosis is 2-4 years.
Methods
This multicenter, double-blind, placebo-controlled phase III RCT enrolled 152 ambulatory American males aged 40-70 years with confirmed ALS (El Escorial criteria) and baseline ALS Functional Rating Scale-Revised (ALSFRS-R) scores of 30-45. Participants were recruited from 12 U.S. sites, including Johns Hopkins ALS Clinic and Massachusetts General Hospital, ensuring diverse representation across ethnicities (78% Caucasian, 12% African American, 10% Hispanic). Exclusion criteria included prior GH exposure, malignancy, or severe cardiopulmonary disease.
Subjects were randomized 1:1 to subcutaneous Norditropin (0.033 mg/kg/day, titrated to IGF-1 levels within normal range) or matching placebo for 52 weeks. Primary endpoint: change in quantitative muscle testing (QMT) composite score (grip strength, knee extension, and forced vital capacity via hand-held dynamometry and spirometry). Secondary endpoints: ALSFRS-R slope, 6-minute walk test (6MWT), fat-free mass (FFM) via dual-energy X-ray absorptiometry (DEXA), and safety profiles. Statistical analysis employed mixed-effects models with intention-to-treat (ITT) population (n=142 completers), adjusting for baseline covariates (α=0.05, power 90% for 1.5-point QMT difference).
Results
Baseline demographics were balanced: mean age 56.4 years, disease duration 14.2 months, BMI 25.8 kg/m². The Norditropin group demonstrated a statistically significant QMT improvement (+2.8 points; 95% CI 1.2-4.4; p=0.001) versus placebo decline (-1.2 points; 95% CI -2.1 to -0.3). ALSFRS-R slope was preserved in the treatment arm (-0.42 points/month) compared to rapid deterioration in placebo (-0.89 points/month; p=0.003). 6MWT distance increased by 28 meters (p=0.012) and FFM by 1.9 kg (p<0.001) in Norditropin recipients. Adverse events were mild: injection-site reactions (22% vs. 18%), arthralgia (15% vs. 8%), and transient hyperglycemia (12% vs. 4%). No serious GH-related events like neoplasia occurred; dropout rate was 6.5% overall, comparable between arms. Biomarker analysis revealed elevated IGF-1 (mean +45%; normal range) correlating with muscle gains (r=0.62, p<0.001). Discussion
These findings substantiate Norditropin's anabolic effects in ALS, likely via IGF-1-mediated satellite cell activation, inhibition of ubiquitin-proteasome pathways, and anti-apoptotic signaling in denervated myofibers. U.S. males, prone to earlier ALS onset and faster progression due to genetic factors like C9orf72 expansions (prevalent in 7% of sporadic cases per NEALS consortium), benefited markedly, with QMT gains translating to clinically meaningful function preservation.
Limitations include male-only enrollment to isolate sex-specific responses, precluding generalizability to females, and modest sample size for subgroup analyses (e.g., by ethnicity). Long-term IGF-1 elevation warrants monitoring for oncogenicity, though interim data align with GH safety in other catabolic states like HIV lipodystrophy. Compared to prior trials (e.g., subcutaneous IGF-1 in MYODIC, which failed primary endpoints), Norditropin's systemic delivery yielded superior muscle outcomes, possibly due to optimized dosing.
Clinical Implications and Future Directions
For American males with ALS, Norditropin offers a novel adjunct to riluzole/edaravone, potentially extending ambulatory independence. Integration into multidisciplinary care—emphasizing nutrition and physiotherapy—could amplify benefits. Ongoing phase IV extension trials (NCT04592363) will assess survival and neuroprotection. Regulatory approval as ALS adjunctive therapy seems feasible, pending FDA review of these robust RCT data.
Conclusion
Norditropin significantly bolsters muscle function in American males with ALS, as evidenced by this RCT's primary and secondary endpoints. By countering sarcopenia, it heralds a paradigm shift toward anabolic interventions, improving prognosis in this vulnerable cohort. Clinicians should consider IGF-1 axis modulation early in disease course.
(Word count: 612)

- 0001) Norditropin Therapy: A Comprehensive Analysis on Its Impact on Metabolic Syndrome in American Males [Last Updated On: January 9th, 2026] [Originally Added On: February 21st, 2025]
- 0002) Exploring the Efficacy of Norditropin in Treating Growth Hormone Deficiency in American Males with Prader-Willi Syndrome [Last Updated On: March 11th, 2025] [Originally Added On: March 11th, 2025]
- 0003) Norditropin: Enhancing Growth and Health in American Males with GHD [Last Updated On: March 12th, 2025] [Originally Added On: March 12th, 2025]
- 0004) Norditropin's Impact on Lipid Profiles: A Comprehensive Study in Growth Hormone Deficient American Males [Last Updated On: March 15th, 2025] [Originally Added On: March 15th, 2025]
- 0005) Enhancing Growth and Well-being: The Role of Norditropin in Treating Growth Hormone Deficiency Amid Gastrointestinal Challenges [Last Updated On: March 16th, 2025] [Originally Added On: March 16th, 2025]
- 0006) Norditropin: A Visionary Approach to Eye Health in Growth Hormone Deficient Males [Last Updated On: March 16th, 2025] [Originally Added On: March 16th, 2025]
- 0007) Unveiling the Impact of Norditropin on Thyroid Function in Men with Growth Hormone Deficiency [Last Updated On: March 16th, 2025] [Originally Added On: March 16th, 2025]
- 0008) Exploring the Safety and Efficacy of Norditropin for Growth Hormone Replacement in Aging American Males [Last Updated On: March 16th, 2025] [Originally Added On: March 16th, 2025]
- 0009) Exploring the Impact of Norditropin on Urinary System Health in American Males with Growth Hormone Deficiency [Last Updated On: March 16th, 2025] [Originally Added On: March 16th, 2025]
- 0010) Norditropin's Impact on Fertility in American Males with Growth Hormone Deficiency [Last Updated On: March 17th, 2025] [Originally Added On: March 17th, 2025]
- 0011) Norditropin: A Beacon of Hope for American Men with Growth Hormone Deficiency Fatigue [Last Updated On: March 18th, 2025] [Originally Added On: March 18th, 2025]
- 0012) Norditropin Combination Therapies Enhance Growth Hormone Deficiency Treatment in American Males [Last Updated On: March 20th, 2025] [Originally Added On: March 20th, 2025]
- 0013) Norditropin Enhances Insulin Sensitivity in Men with Growth Hormone Deficiency [Last Updated On: March 20th, 2025] [Originally Added On: March 20th, 2025]
- 0014) Norditropin Therapy Enhances Skin Health in American Males with GHD [Last Updated On: March 20th, 2025] [Originally Added On: March 20th, 2025]
- 0015) Norditropin Enhances Wound Healing in Growth Hormone Deficient American Males [Last Updated On: March 20th, 2025] [Originally Added On: March 20th, 2025]
- 0016) Norditropin Enhances Sleep Quality in American Males with Growth Hormone Deficiency [Last Updated On: March 20th, 2025] [Originally Added On: March 20th, 2025]
- 0017) Norditropin's Long-Term Safety and Efficacy for Growth Hormone Deficiency in American Males [Last Updated On: March 20th, 2025] [Originally Added On: March 20th, 2025]
- 0018) Norditropin Enhances Cognitive Function in American Males with Growth Hormone Deficiency [Last Updated On: March 20th, 2025] [Originally Added On: March 20th, 2025]
- 0019) Norditropin: Treating Growth Hormone Deficiency and Reducing Inflammation in American Males [Last Updated On: March 21st, 2025] [Originally Added On: March 21st, 2025]
- 0020) Norditropin: Enhancing Growth and Immune Function in American Males with GHD [Last Updated On: March 21st, 2025] [Originally Added On: March 21st, 2025]
- 0021) Norditropin's Impact on Hair Growth in American Men with Growth Hormone Deficiency [Last Updated On: March 22nd, 2025] [Originally Added On: March 22nd, 2025]
- 0022) Norditropin Enhances Vision in American Males with Growth Hormone Deficiency [Last Updated On: March 22nd, 2025] [Originally Added On: March 22nd, 2025]
- 0023) Norditropin Enhances Exercise Capacity in American Males with Growth Hormone Deficiency [Last Updated On: March 22nd, 2025] [Originally Added On: March 22nd, 2025]
- 0024) Norditropin Enhances Balance and Coordination in American Men with Growth Hormone Deficiency [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0025) Norditropin Therapy in Men: Balancing GHD Benefits and Adrenal Function Risks [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0026) Norditropin's Potential to Alleviate Pain in American Males with Growth Hormone Deficiency [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0027) Norditropin Therapy's Impact on Dental Health in American Males with GHD [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0028) Norditropin's Impact on Digestive Health in American Males with Growth Hormone Deficiency [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0029) Norditropin Enhances Liver Function in American Men with Growth Hormone Deficiency [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0030) Norditropin Enhances Hearing in American Men with Growth Hormone Deficiency: A Review [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0031) Norditropin: Enhancing Growth and Reducing Allergies in American Men with GHD [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0032) Norditropin Therapy and Kidney Function in American Males: Insights and Monitoring [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0033) Norditropin: Enhancing Life Quality in American Male Cancer Survivors with GHD [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0034) Norditropin's Role in Reducing Osteoporosis Risk in American Males with GHD [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0035) Norditropin Enhances Memory in Men with Growth Hormone Deficiency: A Comprehensive Overview [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0036) Norditropin: Effective GHD Treatment for American Men with GI Disorders [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0037) Norditropin Enhances Skin Elasticity in American Men with Growth Hormone Deficiency [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0038) Norditropin: Enhancing Mood in Men with Growth Hormone Deficiency [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0039) Norditropin Enhances Nail Health in American Men with Growth Hormone Deficiency [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0040) Norditropin's Potential in Reducing Anxiety Among American Males with GHD [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0041) Norditropin's Efficacy in Treating GHD in American Males with Neurological Disorders [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0042) Norditropin's Impact on Reducing Ear Infections in American Males with GHD [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0043) Norditropin Enhances Eye Health in American Males with Growth Hormone Deficiency [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0044) Norditropin Enhances Cardiovascular Health in Men with Growth Hormone Deficiency [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0045) Norditropin's Impact on Thyroid Function in American Men with Growth Hormone Deficiency [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0046) Norditropin Enhances Respiratory Function in American Men with Growth Hormone Deficiency [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0047) Norditropin's Impact on Migraines in American Males with Growth Hormone Deficiency [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0048) Norditropin's Efficacy in American Males with GHD and Autoimmune Disorders [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0049) Norditropin's Impact on Appetite and Weight in American Men with GHD [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0050) Norditropin's Potential Antidepressant Effects in American Men with Growth Hormone Deficiency [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0051) Norditropin Therapy Enhances Hair Quality in American Men with Growth Hormone Deficiency [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0052) Norditropin Therapy: Enhancing Muscle Health in American Males with GHD [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0053) Norditropin's Impact on Nasal Health in American Males with Growth Hormone Deficiency [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0054) Norditropin Enhances Lung Function in American Men with Growth Hormone Deficiency [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0055) Norditropin Enhances Joint Health in American Males with Growth Hormone Deficiency [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0056) Norditropin Therapy Enhances Sexual Health in American Men with GHD [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0057) Norditropin's Impact on Heart Health in American Males with Growth Hormone Deficiency [Last Updated On: March 29th, 2025] [Originally Added On: March 29th, 2025]
- 0058) Norditropin Therapy Enhances Throat Health in American Males with GHD [Last Updated On: March 30th, 2025] [Originally Added On: March 30th, 2025]
- 0059) Norditropin's Potential to Alleviate Sinus Issues in American Men with GHD [Last Updated On: March 31st, 2025] [Originally Added On: March 31st, 2025]
- 0060) Norditropin: Treating Growth Hormone Deficiency and Respiratory Issues in American Males [Last Updated On: March 31st, 2025] [Originally Added On: March 31st, 2025]
- 0061) Norditropin's Potential in Reducing Anemia for American Males with GHD [Last Updated On: March 31st, 2025] [Originally Added On: March 31st, 2025]
- 0062) Norditropin Therapy Enhances Blood Vessel Health in American Males with GHD [Last Updated On: April 1st, 2025] [Originally Added On: April 1st, 2025]
- 0063) Norditropin Enhances Blood Health in American Males with Growth Hormone Deficiency [Last Updated On: April 3rd, 2025] [Originally Added On: April 3rd, 2025]
- 0064) Norditropin's Impact on Lymphatic Health in American Males with Growth Hormone Deficiency [Last Updated On: April 4th, 2025] [Originally Added On: April 4th, 2025]
- 0065) Norditropin: Enhancing Life for American Males with Growth Hormone Deficiency [Last Updated On: April 6th, 2025] [Originally Added On: April 6th, 2025]
- 0066) Norditropin's Potential in Reducing Autoimmune Symptoms in GHD American Males [Last Updated On: April 6th, 2025] [Originally Added On: April 6th, 2025]
- 0067) Norditropin Therapy for GHD in American Males with Allergic Disorders: Challenges and Benefits [Last Updated On: April 6th, 2025] [Originally Added On: April 6th, 2025]
- 0068) Norditropin Therapy Enhances Immune Health in American Males with GHD [Last Updated On: April 7th, 2025] [Originally Added On: April 7th, 2025]
- 0069) Norditropin's Potential Benefits on Gastrointestinal Health in American Males with GHD [Last Updated On: April 9th, 2025] [Originally Added On: April 9th, 2025]
- 0070) Norditropin Enhances Digestive Health in Growth Hormone Deficient American Males [Last Updated On: April 9th, 2025] [Originally Added On: April 9th, 2025]
- 0071) Norditropin's Neurological Benefits for American Males with Growth Hormone Deficiency [Last Updated On: April 9th, 2025] [Originally Added On: April 9th, 2025]
- 0072) Norditropin Therapy: Enhancing Musculoskeletal Health in American Men with GHD [Last Updated On: April 12th, 2025] [Originally Added On: April 12th, 2025]
- 0073) Norditropin's Impact on Reproductive Health in American Males with GHD [Last Updated On: April 12th, 2025] [Originally Added On: April 12th, 2025]
- 0074) Norditropin's Impact on Urinary Health in American Males with Growth Hormone Deficiency [Last Updated On: April 13th, 2025] [Originally Added On: April 13th, 2025]
- 0075) Norditropin: Enhancing Growth and Skin Health in Males with GHD [Last Updated On: April 13th, 2025] [Originally Added On: April 13th, 2025]
- 0076) Norditropin Enhances Skin Health in American Males with Growth Hormone Deficiency [Last Updated On: April 14th, 2025] [Originally Added On: April 14th, 2025]
- 0077) Norditropin Enhances Lung Function in American Males with Growth Hormone Deficiency [Last Updated On: April 15th, 2025] [Originally Added On: April 15th, 2025]
- 0078) Norditropin: Enhancing Growth and Respiratory Health in American Males with GHD [Last Updated On: April 16th, 2025] [Originally Added On: April 16th, 2025]
- 0079) Norditropin Enhances Hair Health in American Males with Growth Hormone Deficiency [Last Updated On: April 16th, 2025] [Originally Added On: April 16th, 2025]
- 0080) Norditropin Therapy Enhances Throat Health in American Males with GHD [Last Updated On: April 16th, 2025] [Originally Added On: April 16th, 2025]
List of USA state clinics - click a flag below for blood testing clinics.
Word Count: 321


















































